COFEPRIS Medical Device Registration: Step-by-Step Framework for Mexico Market Entry
Strategic, execution-focused COFEPRIS guide for Medtech and Pharma teams entering Mexico: pathway design, timeline realism, cost logic, and risk controls.
.jpeg)
Strategic, execution-focused COFEPRIS guide for Medtech and Pharma teams entering Mexico: pathway design, timeline realism, cost logic, and risk controls.
.jpeg)
Key Takeaways
Monthly insights on COFEPRIS, market access, and compliance changes.
Mexico remains one of the most relevant healthcare growth markets in Latin America, but access is rarely determined by commercial demand alone. For Medtech and Pharma organizations, launch success depends on whether regulatory work is managed as a strategic operating system rather than a late-stage compliance task. In practical terms, COFEPRIS registration determines not only if a product can enter the market, but also how predictable launch timing, partner negotiations, and internal resource planning will be.
This article is written for business development and regulatory teams that need a clearer, execution-driven approach. Instead of overloading each section with checklists, it focuses on decision logic, operating sequence, and the points where delays usually become expensive.
Receive monthly execution-focused updates through the EQ Corporate newsletter for COFEPRIS and Mexico market access teams.
COFEPRIS is Mexico’s federal authority for sanitary risk control, responsible for evaluating and supervising medical devices and other regulated health products before and during commercialization.
Sanitary registration means the formal authorization that enables legal commercialization of a product in Mexico under a defined technical, labeling, and lifecycle-compliance scope.
Regulatory pathway means the sequence of classification, technical preparation, filing, authority exchanges, and post-approval controls needed to sustain compliant operations.
Regulatory readiness means the practical operating condition where evidence quality, role ownership, and decision cadence are strong enough to support authority review without preventable interruption.
For teams validating first principles directly from regulators, COFEPRIS publishes institutional guidance and pathways through its official portal and digital systems, including COFEPRIS official website and DIGIPRiS registration platform.
"The Model recommends guiding principles, harmonized definitions and attributes of effective and efficient regulation to be embodied within binding and enforceable law." — World Health Organization

Before a technical file is assembled, teams should align on intended use, claims scope, and product-risk assumptions. Classification errors at this stage often cascade into inconsistent evidence requests, conflicting label language, and late-cycle corrections. The operational objective is not only to select the right route, but to document the rationale so internal and external stakeholders can defend it consistently through review interactions.
A short requirements matrix usually improves this phase: it maps each expected authority requirement to available evidence status, owners, and remediation deadlines.
Many delays come from dossiers that are complete in volume but inconsistent in logic. COFEPRIS reviewers need technical coherence across indication language, risk controls, quality evidence, and Spanish labeling. When teams treat translation and localization as a final formatting step, terminology drift appears between key artifacts and drives clarification cycles.
For this reason, narrative consistency matters as much as completeness. A high-quality submission explains the product story in one stable logic chain from intended use to quality and safety controls.
Local representation should be addressed as governance architecture, not administrative outsourcing. Roles, escalation rights, decision timeframes, and ownership safeguards need to be explicit before filing begins. This prevents friction when authority observations require rapid, multi-party responses.
Where possible, governance clauses should also define continuity mechanisms so critical regulatory operations remain stable during partner transitions.
Submission quality sets initial momentum, but response quality determines execution speed over the full cycle. Teams that pre-assign observation owners and establish response SLAs generally avoid the internal latency that slows otherwise strong projects. The filing moment should therefore include a response protocol, not only a finalized package.
Typical timing range: broad by product complexity and evidence maturity; use ranges and checkpoints rather than single-date promises.
During authority exchanges, speed without alignment usually creates second-order delays. Effective responses map each authority question to direct evidence and maintain wording consistency across all updated artifacts. This is where cross-functional review discipline protects both timeline and technical credibility.
Teams that run a single observation tracker with accountable owners, due dates, and final approvers consistently perform better than ad hoc email chains.
Approval is a transition point into operating discipline. Post-approval change control, surveillance routines, and renewal planning should already be active before first commercial deployment. Organizations that postpone lifecycle governance often accumulate hidden risk that later disrupts commercial continuity.
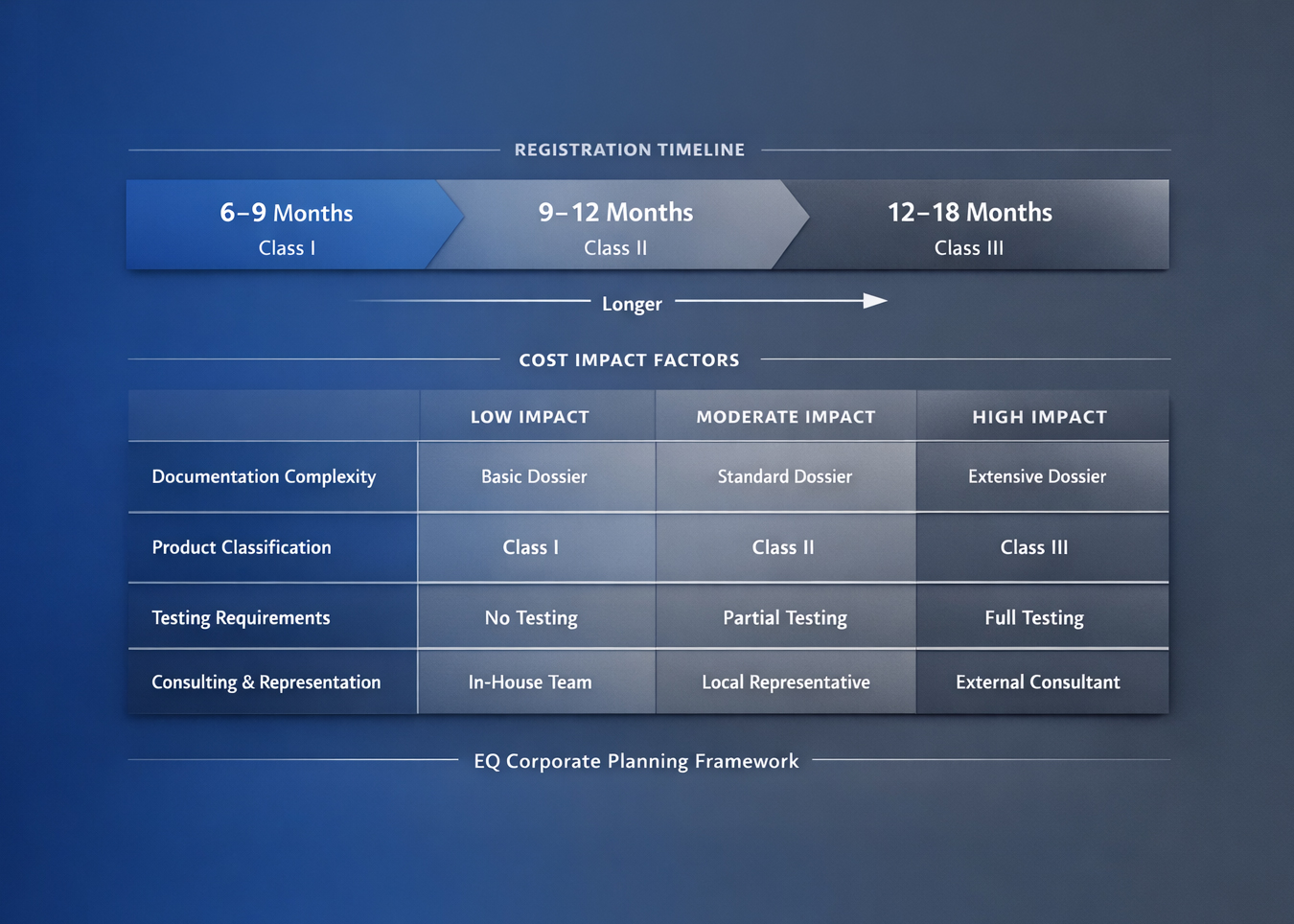
The practical answer is always range-based. Timeline behavior is shaped by product class, dossier maturity, and responsiveness during authority exchanges. A useful planning model separates work into three windows: readiness build, authority interaction, and post-observation closure. This avoids compressing uncertainty into one unrealistic launch date.
From an executive perspective, timeline reliability usually improves when teams define decision rights in advance and keep evidence ownership explicit. Without this governance, projects appear to move quickly at the start, then stall during clarifications.
Cost should be structured as a framework tied to complexity drivers rather than negotiated as a single fixed figure. The strongest drivers are typically technical documentation gaps, classification-specific evidence depth, localization quality requirements, and remediation workload triggered by authority observations.
This is why two products within the same broad category can show materially different cost behavior. Accurate planning requires scenario-based budgeting that includes both baseline submission effort and corrective-cycle contingencies.
"Strategic regulatory governance reduces timeline volatility and improves resource efficiency across market entry programs." — EQ Corporate Insight
Organizations that perform best in COFEPRIS pathways typically share one behavior: they govern regulatory execution as a business-critical operating stream. Swiss precision in preparation, combined with local Mexico execution capability, creates stronger review resilience and more stable launch outcomes.
Across long-horizon market entry programs, predictability comes less from optimistic estimates and more from disciplined process design, evidence traceability, and cross-functional ownership.
Teams needing integrated support can align this pathway with Regulatory Affairs Consulting and connected market-entry planning services.
Book a working session with EQ Corporate to map route assumptions, evidence gaps, governance controls, and launch milestones for Mexico: Book consultation.
COFEPRIS may request clinical or technical evidence aligned with the device risk profile and intended use. In practice, this can include relevant literature, performance documentation, technical reports, and rationale connecting the product claims to supporting evidence. A structured evidence matrix is often useful because it links each claim to its source and helps teams respond faster if clarification is requested. Consistency across labeling, indications, and technical documentation is also important for review efficiency.
COFEPRIS submissions for medical devices usually combine legal-administrative records with technical product evidence. Common components include applicant documentation, product specifications, intended use, manufacturing information, and labeling in the required format. Depending on device classification and pathway, evidence can also include performance testing or clinical support. Many avoidable delays come from inconsistencies between labeling, technical file content, and claimed indications.
COFEPRIS sanitary registration timelines for medical devices in Mexico are often estimated in a 3 to 9 month range. Final timing varies with product risk classification, submission quality, and whether clarification rounds are required. Teams usually reduce avoidable delays by confirming classification early, validating labeling consistency, and preparing technical evidence in a reviewer-friendly structure. Coordinating regulatory milestones with launch planning also helps set realistic expectations across commercial and operations teams.
Most COFEPRIS sanitary registration processes for medical devices are completed in an estimated 3 to 9 month range, but actual timing varies by risk class, submission completeness, and review rounds. Teams generally reduce delays by validating classification early, preparing technical evidence in a clear structure, and checking labeling consistency before filing. A practical approach is to map regulatory milestones and commercial launch dependencies at the same time.
In Mexico, COFEPRIS classification for medical devices is based on risk and intended use. That classification influences the registration pathway, documentation scope, and expected review complexity. Defining class early helps avoid mid-process strategy changes and supports clearer launch planning. Teams typically benefit from validating that labeling, technical specifications, and intended claims all align with the proposed class before submission.
Import permit requirements in Mexico depend on how the product is classified, whether it is already authorized, and the intended commercial or non-commercial use. Some shipments may require specific documentation tied to sanitary registration status, while others follow different mechanisms. Because conditions vary, teams should align import planning with classification and registration strategy early, especially when launch timelines are tight.
Monthly regulatory updates, market access insights, and COFEPRIS process changes curated for medtech and pharma decision-makers.
